Ethanol
| Ethanol | |
|---|---|
| |
| General | |
| Systematic name | Ethanol |
| Other names | Ethyl alcohol, grain alcohol, hydroxyethane, moonshine, drinking alcohol, EtOH |
| Molecular formula | C2H5OH or C2H6O. |
| SMILES | CCO |
| Molar mass | 46.06844(232) g/mol |
| Appearance | colorless clear liquid |
| CAS number | [64-17-5] |
| Properties | |
| Density and phase | 0.789 g/cm³, liquid |
| Solubility in water | Fully miscible |
| Melting point | −114.3 °C (158.8 K) |
| Boiling point | 78.4 °C (351.6 K) |
| Acidity (pKa) | 15.9 (H+ from OH group) |
| Viscosity | 1.200 mPa·s (cP) at 20.0 °C |
| Dipole moment | 5.64 fC·fm (1.69 D) (gas) |
| Hazards | |
| MSDS | External MSDS |
| EU classification | Flammable (F) |
| NFPA 704 | |
| R-phrases | R11 |
| S-phrases | S2, S7, S16 |
| Flash point | 286.15 K (13 °C or 55.4 °F) |
| Flammable limits in air (by volume) |
3.28% - 18.95% |
| RTECS number | KQ6300000 |
| Supplementary data page | |
| Structure & properties | n, εr, etc. |
| Thermodynamic data | Phase behavior Solid, liquid, gas |
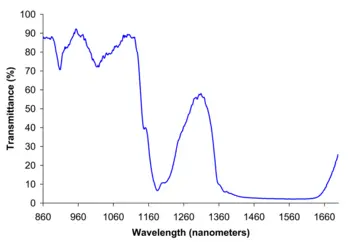
| Spectral data | UV, IR, NMR, MS |
| Related compounds | |
| Related alcohols | Methanol, 1-Propanol |
| Other heteroatoms | Ethylamine, Ethyl chloride, Ethyl bromide, Ethanethiol |
| Substituted ethanols | Ethylene glycol, Ethanolamine, 2-Chloroethanol |
| Other compounds | Acetaldehyde, Acetic acid |
| Except where noted otherwise, data are given for materials in their standard state (at 25°C, 100 kPa) | |
Ethanol, also known as ethyl alcohol, drinking alcohol, or grain alcohol, is a flammable, colorless, slightly toxic chemical compound with a distinctive perfume-like odor. It is best known as the alcohol found in alcoholic beverages. In common usage, it is often referred to simply as alcohol. Its molecular formula is variously represented as EtOH, CH3CH2OH, C2H5OH or C2H6O.
Physical properties
Ethanol's hydroxyl group is able to participate in hydrogen bonding. At the molecular level, liquid ethanol consists of hydrogen-bonded pairs of ethanol molecules; this phenomenon renders ethanol more viscous and less volatile than less polar organic compounds of similar molecular weight. In the vapor phase, there is little hydrogen bonding; ethanol vapor consists of individual ethanol molecules. Ethanol, like most short-chain alcohols, is flammable, colorless, has a strong odor, and is volatile.
Ethanol has a refractive index of 1.3614. Ethanol is a versatile solvent. It is miscible with water and with most organic liquids, including nonpolar liquids such as aliphatic hydrocarbons. Organic solids of low molecular weight are usually soluble in ethanol. Among ionic compounds, many monovalent salts are at least somewhat soluble in ethanol, with salts of large, polarizable ions being more soluble than salts of smaller ions. Most salts of polyvalent ions are practically insoluble in ethanol.
Furthermore, ethanol is used as a solvent in dissolving medicines, food flavorings and colorings that do not dissolve easily in water. Once the non-polar material is dissolved in the ethanol, water can be added to prepare a solution that is mostly water. The ethanol molecule has a hydrophilic -OH group that helps it dissolve polar molecules and ionic substances. The short, hydrophobic hydrocarbon chain CH3CH2- can attract non-polar molecules. Thus, ethanol can dissolve both polar and non-polar substances.
Several unusual phenomena are associated with mixtures of ethanol and water. Ethanol-water mixtures have less volume than their individual components: a mixture of equal volumes ethanol and water has only 95.6 percent of the volume of equal parts ethanol and water, unmixed. The addition of even a small amount of ethanol to water sharply reduces the surface tension of water. This property partially explains the tears of wine phenomenon: when wine is swirled inside a glass, ethanol evaporates quickly from the thin film of wine on the wall of the glass. As its ethanol content decreases, its surface tension increases, and the thin film beads up and runs down the glass in channels rather than as a smooth sheet.
Chemistry
The chemistry of ethanol is largely that of its hydroxyl group.
- Acid-base chemistry
Ethanol's hydroxyl proton is weakly acidic, having a pKa of only 15.9, compared to water's 15.7[1] (Ka of ethanol is a measure of . Note that Ka of water is derived by dividing water's dissociation constant, moles2/liter, by its molar density of 55.5 moles/liter). Ethanol can be quantitatively converted to its conjugate base, the ethoxide ion (CH3CH2O−), by reaction with an alkali metal such as sodium. This reaction evolves hydrogen gas:
- Nucleophilic substitution
In aprotic solvents, ethanol reacts with hydrogen halides to produce ethyl halides such as ethyl chloride and ethyl bromide via nucleophilic substitution:
Ethyl halides can also be produced by reacting ethanol by more specialized halogenating agents, such as thionyl chloride for preparing ethyl chloride, or phosphorus tribromide for preparing ethyl bromide.
- Esterification
Under acid-catalysed conditions, ethanol reacts with carboxylic acids to produce ethyl esters and water:
- RCOOH + HOCH2CH3 → RCOOCH2CH3 + H2O
The reverse reaction, hydrolysis of the resulting ester back to ethanol and the carboxylic acid, limits the extent of reaction, and high yields are unusual unless water can be removed from the reaction mixture as it is formed. Esterification can also be carried out using more a reactive derivative of the carboxylic acid, such as an acyl chloride or acid anhydride. A very common ester of ethanol is ethyl acetate, found in for example nail polish remover.
Ethanol can also form esters with inorganic acids. Diethyl sulfate and triethyl phosphate, prepared by reacting ethanol with sulfuric and phosphoric acid, respectively, are both useful ethylating agents in organic synthesis. Ethyl nitrite, prepared from the reaction of ethanol with sodium nitrite and sulfuric acid, was formerly a widely-used diuretic.
- Dehydration
Strong acids, such as sulfuric acid, can catalyse ethanol's dehydration to form either diethyl ether or ethylene:
- 2 CH3CH2OH → CH3CH2OCH2CH3 + H2O
Although sulfuric acid catalyses this reaction, the acid is diluted by the water that is formed, which makes the reaction inefficient. Which product, diethyl ether or ethylene, predominates depends on the precise reaction conditions.
- Oxidation
Ethanol can be oxidized to acetaldehyde, and further oxidized to acetic acid. In the human body, these oxidation reactions are catalysed by enzymes. In the laboratory, aqueous solutions of strong oxidizing agents, such as chromic acid or potassium permanganate, oxidize ethanol to acetic acid, and it is difficult to stop the reaction at acetaldehyde at high yield. Ethanol can be oxidized to acetaldehyde, without overoxidation to acetic acid, by reacting it with pyridinium chromic chloride.
- Combustion
Combustion of ethanol forms carbon dioxide and water:
- C2H5OH + 3 O2 → 2 CO2 + 3 H2O
Production
Ethanol is produced both as a petrochemical, through the hydration of ethylene, and biologically, by fermenting sugars with yeast.
Ethylene hydration
Ethanol for use as industrial feedstock is most often made from petrochemical feedstocks, typically by the acid-catalyzed hydration of ethene, represented by the chemical equation
The catalyst is most commonly phosphoric acid, adsorbed onto a porous support such as diatomaceous earth or charcoal; this catalyst was first used for large-scale ethanol production by the Shell Oil Company in 1947.[2] Solid catalysts, mostly various metal oxides, have also been mentioned in the chemical literature.
In an older process, first practiced on the industrial scale in 1930 by Union Carbide,[2] but now almost entirely obsolete, ethene was hydrated indirectly by reacting it with concentrated sulfuric acid to produce ethyl sulfate, which was then hydrolysed to yield ethanol and regenerate the sulfuric acid:
- C2H4 + H2SO4 → CH3CH2SO4H
- CH3CH2SO4H + H2O → CH3CH2OH + H2SO4
Fermentation
Ethanol for use in alcoholic beverages, and the vast majority of ethanol for use as fuel, is produced by fermentation: when certain species of yeast (most importantly, Saccharomyces cerevisiae) metabolize sugar in the absence of oxygen, they produce ethanol and carbon dioxide. The overall chemical reaction conducted by the yeast may be represented by the chemical equation
The process of culturing yeast under conditions to produce alcohol is referred to as brewing. Brewing can only produce relatively dilute concentrations of ethanol in water; concentrated ethanol solutions are toxic to yeast. The most ethanol-tolerant strains of yeast can survive in up to about 15 percent ethanol (by volume).
During the fermentation process, it is important to prevent oxygen from getting to the ethanol, since otherwise the ethanol would be oxidised to acetic acid (vinegar). Also, in the presence of oxygen, the yeast would undergo aerobic respiration to produce just carbon dioxide and water, without producing ethanol.
In order to produce ethanol from starchy materials such as cereal grains, the starch must first be broken down into sugars. In brewing beer, this has traditionally been accomplished allowing the grain to germinate, or malt. In the process of germination, the seed produces enzymes that can break its starches into sugars. For fuel ethanol, this hydrolysis of starch into glucose is accomplished more rapidly by treatment with dilute sulfuric acid, fungal amylase enzymes, or some combination of the two.
Feedstocks
Currently the main feedstock in the United States for the production of ethanol is corn. Approximately 2.8 gallons of ethanol (10 liters) are produced from one bushel of corn (35 liters). While much of the corn turns into ethanol, some of the corn also yields by-products such as DDGS (distillers dried grains with solubles) that can be used to fulfill a portion of the diet of livestock. A bushel of corn produces about 18 pounds of DDGS.[3] Critics of ethanol as fuel decry the use of corn to produce ethanol because corn is an energy-intensive crop that requires petroleum-derived fertilizers; however, using corn to produce alcohol could save farmers additional petroleum if the farmers are feeding the byproduct to livestock and if the excrement from the animals is then used as fertilizer for the corn.[4] Although most of the fermentation plants have been built in corn-producing regions, sorghum is also an important feedstock for ethanol production in the Plains states. Pearl millet is showing promise as an ethanol feedstock for the southeastern United States
In some parts of Europe, particularly France and Italy, wine is used as a feedstock due to massive oversupply.[5] Japan is hoping to use rice wine (sake) as an ethanol source.[6]
At petroleum prices like those that prevailed through much of the 1990s, ethylene hydration was a decidedly more economical process than fermentation for producing purified ethanol. Later increases in petroleum prices, coupled with perennial uncertainty in agricultural prices, make forecasting the relative production costs of fermented versus petrochemical ethanol difficult.
Testing
In breweries and biofuel plants, the quantity of ethanol present is measured using one of two methods. Infrared ethanol sensors measure the vibrational frequency of dissolved ethanol using the CH band at 2900cm-1. This method uses a relatively inexpensive solid state sensor that compares the CH band with a reference band to calculate the ethanol content. This calculation makes use of the Beer-Lambert law.
Alternatively, by measuring the density of the starting material, and the density of the product, using a hydrometer, the change in gravity during fermentation is used to derive the alcohol content. This is an inexpensive and indirect method but has a long history in the beer brewing industry.
Purification
The product of either ethylene hydration or brewing is an ethanol-water mixture. For most industrial and fuel uses, the ethanol must be purified. Fractional distillation can concentrate ethanol to 95.6 percent by weight (89.5 mole percent). The mixture of 95.6 percent ethanol and 4.4 percent water (percentage by weight) is an azeotrope with a boiling point of 78.2 °C, and cannot be further purified by distillation. Therefore, 95 percent ethanol in water is a fairly common solvent.
After distillation ethanol can be further purified by "drying" it using lime or salt. When lime (calcium oxide) is mixed with the water in ethanol, calcium hydroxide forms. The calcium hydroxide can then be separated from the ethanol. Dry salt will dissolve some of the water content of the ethanol as it passes through, leaving a purer alcohol.[7]
Several approaches are used to produce absolute ethanol. The ethanol-water azeotrope can be broken by the addition of a small quantity of benzene. Benzene, ethanol, and water form a ternary azeotrope with a boiling point of 64.9 °C. Since this azeotrope is more volatile than the ethanol-water azeotrope, it can be fractionally distilled out of the ethanol-water mixture, extracting essentially all of the water in the process. The bottoms from such a distillation is anhydrous ethanol, with several parts per million residual benzene. Benzene is toxic to humans, and cyclohexane has largely supplanted benzene in its role as the entrainer in this process.
Alternatively, a molecular sieve can be used to selectively absorb the water from the 95.6 percent ethanol solution. Synthetic zeolite in pellet form can be used, as well as a variety of plant-derived absorbents, including cornmeal, straw, and sawdust. The zeolite bed can be regenerated essentially an unlimited number of times by drying it with a blast of hot carbon dioxide. Cornmeal and other plant-derived absorbents cannot readily be regenerated, but where ethanol is made from grain, they are often available at low cost. Absolute ethanol produced this way has no residual benzene, and can be used to fortify port and sherry in traditional winery operations. Membranes can also be used to separate ethanol and water. The membrane can break the water-ethanol azeotrope because separation is not based on vapor-liquid equilibria. Membranes are often used in the so-called hybrid membrane distillation process. This process uses a pre-concentration distillation column as first separating step. The further separation is then accomplished with a membrane operated either in vapor permeation or pervaporation mode. Vapor permeation uses a vapor membrane feed and pervaporation uses a liquid membrane feed.
At pressures less than atmospheric pressure, the composition of the ethanol-water azeotrope shifts to more ethanol-rich mixtures, and at pressures less than 70 torr (9.333 kPa) , there is no azeotrope, and it is possible to distill absolute ethanol from an ethanol-water mixture. While vacuum distillation of ethanol is not presently economical, pressure-swing distillation is a topic of current research. In this technique, a reduced-pressure distillation first yields an ethanol-water mixture of more than 95.6 percent ethanol. Then, fractional distillation of this mixture at atmospheric pressure distills off the 95.6 percent azeotrope, leaving anhydrous ethanol at the bottoms.
Prospective technologies
Glucose for fermentation into ethanol can also be obtained from cellulose. Until recently, however, the cost of the cellulase enzymes that could hydrolyse cellulose has been prohibitive. The Canadian firm Iogen brought the first cellulose-based ethanol plant on-stream in 2004.[8] The primary consumer thus far has been the Canadian government, which, along with the United States government (particularly the Department of Energy's National Renewable Energy Laboratory), has invested millions of dollars into assisting the commercialization of cellulosic ethanol. Realization of this technology would turn a number of cellulose-containing agricultural byproducts, such as corncobs, straw, and sawdust, into renewable energy resources.
Other enzyme companies are developing genetically engineered fungi which would produce large volumes of cellulase, xylanase and hemicellulase enzymes which can be utilized to convert agricultural residues such as corn stover, distiller grains, wheat straw and sugar cane bagasse and energy crops such as Switchgrass into fermentable sugars which may be used to produce cellulosic ethanol. [9]
Cellulosic materials typically contain, in addition to cellulose, other polysaccharides, including hemicellulose. When hydrolysed, hemicellulose breaks down into mostly five-carbon sugars such as xylose. S. cerevisiae, the yeast most commonly used for ethanol production, cannot metabolize xylose. Other yeasts and bacteria are under investigation to metabolize xylose and so improve the ethanol yield from cellulosic material.[10]
The anaerobic bacterium Clostridium ljungdahlii, recently discovered in commercial chicken wastes, can produce ethanol from single-carbon sources including synthesis gas, a mixture of carbon monoxide and hydrogen that can be generated from the partial combustion of either fossil fuels or biomass. Use of these bacteria to produce ethanol from synthesis gas has progressed to the pilot plant stage at the BRI Energy facility in Fayetteville, Arkansas.[11]
Another prospective technology is the closed-loop ethanol plant. Ethanol produced from corn has a number of critics who suggest that it is primarily just recycled fossil fuels because of the energy required to grow the grain and convert it into ethanol. However, the closed-loop ethanol plant attempts to address this criticism. In a closed-loop plant, the energy for the distillation comes from fermented manure, produced from cattle that have been fed the by-products from the distillation. The leftover manure is then used to fertilize the soil used to grow the grain. Such a process is expected to have a much lower fossil fuel requirement.[12] However, general thermodynamic considerations indicate that the total efficiency of such plants, in combination with the production of cellulose/sugar, will remain relatively low.
Types of ethanol
Denatured alcohol
In most jurisdictions, the sale of ethanol, as a pure substance or in the form of alcoholic beverages, is heavily taxed. In order to relieve non-beverage industries of this tax burden, governments specify formulations for denatured alcohol, which consists of ethanol blended with various additives to render it unfit for human consumption. These additives, called denaturants, are generally either toxic (such as methanol) or have unpleasant tastes or odors (such as denatonium benzoate).
Specialty denatured alcohols are denatured alcohol formulations intended for a particular industrial use, containing denaturants chosen so as not to interfere with that use. While they are not taxed, purchasers of specialty denatured alcohols must have a government-issued permit for the particular formulation they use and must comply with other regulations.
Completely denatured alcohols are formulations that can be purchased for any legal purpose, without permit, bond, or other regulatory compliance. It is intended that it be difficult to isolate a product fit for human consumption from completely denatured alcohol. For example, the completely denatured alcohol formulation used in the United Kingdom contains (by volume) 89.66 percent ethanol, 9.46 percent methanol, 0.50 percent pyridine, 0.38 percent naphtha, and is dyed purple with methyl violet.[13]
Absolute ethanol
Absolute or anhydrous alcohol generally refers to purified ethanol, containing no more than one percent water.
It is not possible to obtain absolute alcohol by simple fractional distillation, because a mixture containing around 95.6 percent alcohol and 4.4 percent water becomes a constant boiling mixture (an azeotropic mixture). In one common industrial method to obtain absolute alcohol, a small quantity of benzene is added to rectified spirit and the mixture is then distilled. Absolute alcohol is obtained in the third fraction that distills over at 78.2 °C (351.3 K).
Because a small amount of the benzene used remains in the solution, absolute alcohol produced by this method is not suitable for consumption as benzene is carcinogenic.
There is also an absolute alcohol production process by desiccation using glycerol. Alcohol produced by this method is known as spectroscopic alcohol—so called because the absence of benzene makes it suitable as a solvent in spectroscopy.
Currently, the most popular method of purification past 95.6 percent purity is desiccation using adsorbents such as starch or zeolites, which adsorb water preferentially. Azeotropic distillation and extractive distillation techniques also exist.
Pure ethanol is classed as 200 proof in the United States, equivalent to 175 degrees proof in the (now rarely used) UK system.
Neutralized ethanol
Neutralized ethanol is used for some analytical purposes. The pH indicators are acid/base molecules that change their color requiring certain amount of acid or base. Neutralized ethanol is used in order to compensate for this error. The indicator (phenolphthalein, for example) is added to the ethanol solvent first and KOH is added until the color of the solution turns pale pink. The so obtained "neutralized ethanol" is then added to the target of the titration, which may be sample of neat organic acid. The titration stops when the same pale pink color is achieved. This way, the indicator neutralization error is eliminated.
Use
As a fuel

The largest single use of ethanol is as a motor fuel and fuel additive. The largest national fuel ethanol industries exist in Brazil (gasoline sold in Brazil contains at least 20 percent ethanol and hydrous ethanol is also used as fuel).[14] In order for ethanol to be suitable for use as a replacement to petrol in its pure form, it must be distilled to at least 70-80 percent purity by volume before use. For use as an additive to petrol, almost all water must be removed, otherwise it will separate from the mixture and settle to the bottom of the fuel tank, causing the fuel pump to draw water into the engine, which will cause the engine to stall.[15]
Today almost 50 percent of Brazilian cars are able to use 100 percent ethanol as fuel, that includes ethanol only engines and flex fuel engines. Flex fuel engines are able to work with all ethanol, all gasoline or any mixture of both, giving the buyer a choice for a perfect balance between price/performance issue. That was only possible due to the capability of an efficient sugar cane production. Sugar cane not only has a greater concentration of sucrose (about 30 percent more than corn) but is also much easier to extract. The bagasse generated by the process is not wasted and it is utilized in power plants becoming a surprisingly efficient source of electricity. World production of ethanol in 2015 was 24.7 billion gallons, with 88 percent of the world supply coming from Brazil and the United States.[16]
One method of production is through fermentation of sugar. Ethanol creates very little pollution when burned. Millions more acres of land are needed if ethanol is to be used to replace gasoline. Pure ethanol has a lower energy content than gasoline (about 30 percent less energy per unit volume). At gas stations, ethanol is contained in a mix of ethanol and gasoline, otherwise known as gasohol. In the United States, the color yellow (symbolizing the color of corn) has become associated with the fuel and is commonly used on fuel pumps and labels. The US uses Gasohol (max 10 percent ethanol) and E85 (85 percent ethanol) ethanol/gasoline mixtures.
Controversy
As reported in "The Energy Balance of Corn Ethanol: an Update,"[17] the energy returned on energy invested (EROEI) for ethanol made from corn in the United States is 1.34 (it yields 34 percent more energy than it takes to produce it). Input energy includes natural gas based fertilizers, farm equipment, transformation from corn or other materials, and transportation.
Oil has historically had a much higher EROEI, especially on land in areas with pressure support, but also under the sea, which only offshore drilling rigs can get to. Apart from this, the amount of ethanol needed to run the United States, for example, is greater than its own farmland could produce, even if fields used for food were converted into cornfields. For these reasons, many people do not view ethanol alone as a solution to replacing conventional oil. Others, however, disagree, pointing out that ethanol production does not necessarily have to come from the farming of corn. For instance, Liquid Fuels of Ohio produces ethanol from expired groceries.
Politics has played a significant role in this issue. Advocates for wheat, corn, and sugar growers have succeeded in their attempts to lobby for regulatory intervention encouraging adoption of ethanol,[18] stimulating debate over who the major beneficiaries of increased use of ethanol would be. Some researchers have warned that ethanol produced from agricultural feedstocks will cause a global food shortage, contributing to starvation in the Third World.[19]
This has led to the development of alternative production methods that use feedstocks such as municipal waste or recycled products, rice hulls, sugarcane bagasse, small diameter trees, wood chips, and switchgrass. These methods have not yet reached the stage of commercialization.
Research shows that fuel consumption increases with the concentration of ethanol in a fuel blend. An Australian study concluded that a 10 percent ethanol blend (E10) yielded a 2.6-2.8 percent increase in consumption.
Blends of up to 10 percent are normally regarded as the safe maximum for a vehicle designed to operate on petroleum. However ethanol blends can run at up to 85 percent or higher in specially designed flexible fueled vehicles.
Consumer Reports, October 2006, questions the fuel economy of a flex fuel vehicle [6]. Specifically, the report notes that fuel economy drops when an automobile uses E-85.
Rocket Fuel
Ethanol has been used as fuel in bipropellant rocket vehicles, in conjunction with an oxidizer. For example, the German V-2 rocket of World War 2 used ethanol fuel.
Alcoholic beverages
Alcoholic beverages vary considerably in their ethanol content and in the foodstuffs from which they are produced. Most alcoholic beverages can be broadly classified as fermented beverages, beverages made by the action of yeast on sugary foodstuffs, or as distilled beverages, beverages whose preparation involves concentrating the ethanol in fermented beverages by distillation. The ethanol content of a beverage is usually measured in terms of the volume fraction of ethanol in the beverage, expressed either as a percentage or in alcoholic proof units.
Fermented beverages can be broadly classified by the foodstuff from which they are fermented. Beers are made from cereal grains or other starchy materials, wines and ciders from fruit juices, and meads from honey. Cultures around the world have made fermented beverages from numerous other foodstuffs, and local and national names for various fermented beverages abound. Fermented beverages may contain up to 15–25 percent ethanol by volume, the upper limit being set by the yeast's tolerance for ethanol, or by the amount of sugar in the starting material.
Distilled beverages are made by distilling fermented beverages. Broad categories of distilled beverages include whiskeys, distilled from fermented cereal grains; brandies, distilled from fermented fruit juices, and rum, distilled from fermented molasses or sugarcane juice. Vodka and similar neutral grain spirits can be distilled from any fermented material (grain or potatoes are most common); these spirits are so thoroughly distilled that no tastes from the particular starting material remain. Numerous other spirits and liqueurs are prepared by infusing flavors from fruits, herbs, and spices into distilled spirits. A traditional example is gin, the infusion of juniper berries into neutral grain alcohol.
In a few beverages, ethanol is concentrated by means other than distillation. Applejack is traditionally made by freeze distillation: water is frozen out of fermented apple cider, leaving a more ethanol-rich liquid behind. Eisbier (most commonly, eisbock) is also freeze-distilled, with beer as the base beverage. Fortified wines are prepared by adding brandy or some other distilled spirit to partially-fermented wine. This kills the yeast and conserves some of the sugar in grape juice; such beverages are not only more ethanol-rich, but are often sweeter than other wines.
Alcoholic beverages are sometimes added to food in cooking, not only for their inherent flavors, but also because the alcohol dissolves flavor compounds that water cannot.
Chemicals derived from ethanol
- Ethyl esters
In the presence of an acid catalyst (typically sulfuric acid) ethanol reacts with carboxylic acids to produce ethyl esters:
The two largest-volume ethyl esters are ethyl acrylate (from ethanol and acrylic acid) and ethyl acetate (from ethanol and acetic acid). Ethyl acrylate is a monomer used to prepare acrylate polymers for use in coatings and adhesives. Ethyl acetate is a common solvent used in paints, coatings, and in the pharmaceutical industry; its most familiar application in the household is as a solvent for nail polish. A variety of other ethyl esters are used in much smaller volumes as artificial fruit flavorings.
- Vinegar
Vinegar is a dilute solution of acetic acid prepared by the action of Acetobacter bacteria on ethanol solutions. Although traditionally prepared from alcoholic beverages including wine, apple cider, and unhopped beer, vinegar can also be made from solutions of industrial ethanol. Vinegar made from distilled ethanol is called "distilled vinegar," and is commonly used in food pickling and as a condiment.
- Ethylamines
When heated to 150–220 °C over a silica- or alumina-supported nickel catalyst, ethanol and ammonia react to produce ethylamine. Further reaction leads to diethylamine and triethylamine:
- CH3CH2OH + NH3 → CH3CH2NH2 + H2O
- CH3CH2OH + CH3CH2NH2 → (CH3CH2)2NH + H2O
- CH3CH2OH + (CH3CH2)2NH → (CH3CH2)3N + H2O
The ethylamines find use in the synthesis of pharmaceuticals, agricultural chemicals, and surfactants.
- Other chemicals
Ethanol in the past has been used commercially to synthesize dozens of other high-volume chemical commodities. At the present, it has been supplanted in many applications by less costly petrochemical feedstocks. However, in markets with abundant agricultural products, but a less developed petrochemical infrastructure, such as the People's Republic of China, Pakistan, India, and Brazil, ethanol can be used to produce chemicals that would be produced from petroleum in the West, including ethylene and butadiene.
Other uses
Ethanol is easily soluble in water in all proportions with a slight overall decrease in volume when the two are mixed. Absolute ethanol and 95 percent ethanol themselves are good solvents, somewhat less polar than water and used in perfumes, paints and tinctures. Other proportions of ethanol with water or other solvents can also be used as a solvent. Alcoholic drinks have a large variety of tastes because various flavor compounds are dissolved during brewing. When ethanol is produced as a mixing beverage it is a neutral grain spirit.
Ethanol is used in medical wipes and in most common antibacterial hand sanitizer gels at a concentration of about 62 percent (percentage by weight, not volume) as an antiseptic. The peak of the disinfecting power occurs around 70 percent ethanol; stronger and weaker solutions of ethanol have a lessened ability to disinfect. Solutions of this strength are often used in laboratories for disinfecting work surfaces. Ethanol kills organisms by denaturing their proteins and dissolving their lipids and is effective against most bacteria and fungi, and many viruses, but is ineffective against bacterial spores. Alcohol does not act like an antibiotic and is not effective against infections by ingestion. Ethanol in the low concentrations typically found in most alcoholic beverages does not have useful disinfectant or antiseptic properties, internally or externally. Ethanol is often used as an antidote in cases of methanol poisoning.
Wine with less than 16 percent ethanol is vulnerable to bacteria. Because of this, port is often fortified with ethanol to at least 18 percent ethanol by volume to halt fermentation. This helps retain sweetness, and in preparation for aging it becomes possible to prevent the invasion of bacteria into the port, and to store the port for long periods of time in wooden containers that can 'breathe', thereby permitting the port to age safely without spoiling. Because of ethanol's disinfectant property, alcoholic beverages of 18 percent ethanol or more by volume can be safely stored for a very long time.
Ethanol is also used in design and sketch art markers, such as Copic, and Tria.
Metabolism and toxicology
Pure ethanol is a tasteless liquid with a strong and distinctive odor that produces a characteristic heat-like sensation when brought into contact with the tongue or mucous membranes. When applied to open wounds (as for disinfection) it produces a strong stinging sensation. Pure or highly concentrated ethanol may permanently damage living tissue on contact. Ethanol applied to unbroken skin cools the skin rapidly through evaporation.
Metabolism
In the human body, ethanol is first oxidized to acetaldehyde, then to acetic acid. The first step is catalyzed by the enzyme alcohol dehydrogenase, and the second by acetaldehyde dehydrogenase.
Magnitude of effect
Some individuals have less effective forms of one or both of these enzymes, and can experience more severe symptoms from ethanol consumption than others. Conversely, those who have acquired ethanol tolerance have a greater quantity of these enzymes, and metabolize ethanol more rapidly.
| BAC (mg/dL) | Symptoms[20] |
|---|---|
| 50 | Euphoria, talkativeness, relaxation |
| 100 | Central nervous system depression, impaired motor and sensory function, impaired cognition |
| >140 | Decreased blood flow to brain |
| 300 | Stupefaction, possible unconsciousness |
| 400 | Possible death |
| >550 | Expiration |
The amount of ethanol in the body is typically quantified by blood alcohol content (BAC), the milligrams of ethanol per 100 milliliters of blood. The table at right summarizes the symptoms of ethanol consumption. Small doses of ethanol generally produce euphoria and relaxation; people experiencing these symptoms tend to become talkative and less inhibited, and may exhibit poor judgment. At higher dosages (BAC > 100mg/dl), ethanol acts as a central nervous system depressant, producing at (progressively higher dosages) impaired sensory and motor function, slowed cognition, stupefaction, unconsciousness, and possible death.
Acetaldehyde toxicology
The initial product of ethanol metabolism, acetaldehyde, is more toxic than ethanol itself. The body can quickly detoxify some acetaldehyde by reaction with glutathione and similar thiol-containing biomolecules. When acetaldehyde is produced beyond the capacity of the body's glutathione supply to detoxify it, it accumulates in the bloodstream until further oxidized to acetic acid. The headache, nausea, and malaise associated with an alcohol hangover stem from a combination of dehydration and acetaldehyde poisoning; many health conditions associated with chronic ethanol abuse, including liver cirrhosis, alcoholism, and some forms of cancer, have been linked to acetaldehyde. The judicial system in the United States, in a number of jurisdictions, controversially, promoted the use of disulfiram, known as Antabuse, for persons convicted of driving while (alcohol) intoxicated. Disulfiram interferes with hepatic acetaldehyde metabolism, exacerbating the discomforts noted above. Some medications, including paracetamol (acetaminophen), as well as exposure to organochlorides, can deplete the body's glutathione supply, enhancing both the acute and long-term risks of even moderate ethanol consumption. Frequent use of alcoholic beverages has also been shown to be a major contributing factor in cases of elevated blood levels of triglycerides.[21]
Bacteria-favoring
Ethanol has been shown to increase the growth of Acinetobacter baumannii, a bacterium responsible for pneumonia, meningitis and urinary tract infections. This finding may contradict the common misconception that drinking alcohol could kill off a budding infection.[22]
Detection
A) Preliminary Test
- Add to 5 mL of sample (Ethyl Alcohol) 2 drops of Potassium dichromate or Potassium Permanganate and an equal amount of concentrated Sulfuric acid, then boil. Sample is positive for ethanol when the dichromate or permanganate is reduced, and the odor of acetaldehyde develops.
B) Lieben's Iodoform Test
- Warm 3 to 5 mL of sample (Ethyl Alcohol) with 1 to 3 mL of strong solution of iodine (Lugol's Solution). Add Potassium Hydroxide solution until the color is faintly yellow. A precipitate of iodoform is formed. The other primary alcohol, aldehydes and ketones, ethyl acetate and lactic acid, also give positive results . The Iodoform crystal may be identified under the microscope appearing as hexagonal plates and stars.
C) Vitali Reaction
- With 1 mL of sample (Ethyl Alcohol), add 3 drops of Carbon disulfide and a pellet of Potassium hydroxide in a small evaporating dish. Heat and when most of the carbon disulfide has evaporated, add 1 drop of Ammonium molybdate solution (1:10). Acidify with concentrated Sulfuric acid. Positive results gives a violet-colored solution. Acetaldehyde and Acetone react similarly and would yield the positive result.
Hazards
- Ethanol-water solutions greater than about 50 percent ethanol by volume are flammable (in some cases ethanol will burn at as low as a 45 percent solution) and easily ignited. Ethanol-water solutions below 50 percent ethanol by volume may also be flammable if the solution is vaporized by heating (as in some cooking methods that call for wine to be added to a hot pan, causing it to flash boil into a vapor, which is then ignited to "burn off" excessive alcohol).
See also
Notes
- ↑ Andrew Streitweiser Jr., and Clayton H. Heathcock, 1976, Introduction to Organic Chemistry. Macmillan. 215
- ↑ 2.0 2.1 J.E. Lodgsdon, "Ethanol." In J.I. Kroschwitz (ed.), Encyclopedia of Chemical Technology, 4th ed. New York: John Wiley & Sons, 1994.
- ↑ Frequently Asked Questions. Red Trail Energy, LLC.
- ↑ Lynn Ellen Doxon, The Alcohol Fuel Handbook (Tallgrass Research Center, 1980).
- ↑ Caroline Wyatt, Draining France's 'wine lake'. BBC news, August 10, 2006. Retrieved October 19,2016.
- ↑ Steve Inskeep, Japan Plans Its Own Green Fuel. Morning Edition, NPR, May 15, 2007. Retrieved October 19, 2016.
- ↑ S.W. Mathewson, The Manual for the Home and Farm Production of Alcohol Fuel (Berkeley, CA: Ten Speed Press, 1980, ISBN 0898150302). Retrieved October 19, 2016.
- ↑ S.K. Ritter, "Biomass or Bust." Chemical & Engineering News 82(22) (2004):31–34.
- ↑ P.C. Badger, Ethanol From Cellulose: A General Review Retrieved October 19, 2016. In: J. Janick and A. Whipkey (eds.), Trends in new crops and new uses (Alexandria, VA: ASHS Press, 2002).
- ↑ Nicolae Sfetcu, Health & Drugs: Disease, Prescription & Medication (Lulu, 2014, ISBN 978-1312039995).
- ↑ Sustainable Energy Resources. Bioengineering Resources, Inc. Retrieved October 19, 2016.
- ↑ R. Rapier, "E3 Biofuels: Responsible Ethanol" R-Squared Energy Blog, 2006. Retrieved October 19, 2106.
- ↑ Great Britain, The Denatured Alcohol Regulations 2005. Statutory Instrument 2005 No. 1524. Retrieved October 19, 2016.
- ↑ Monte Reel, "Brazil's Road to Energy Independence" The Washington Post, August 20, 2006. Retrieved October 19, 2016.
- ↑ Gasohol, E-85, and pure alcohol vs. "Make your own". Retrieved October 19, 2016.
- ↑ Renewable Fuels Association Industry Statistics. Retrieved October 19, 2016.
- ↑ Hosein Shapouri, James A. Duffield, and Michael Wang, The Energy Balance of Corn Ethanol: an Update. United States Department of Agriculture, June 1, 2002.
- ↑ U.S. seeks to boost ethanol, biodiesel. USA Today, April 11, 2007. Retrieved October 19, 2016.
- ↑ U.N.: Not so fast with ethanol, other biofuels. Associated Press, May 8, 2007. Retrieved October 19, 2016.
- ↑ L.A. Pohorecky and J. Brick, 1988, "Pharmacology of ethanol." Pharmacology & Therapeutics 36(3):335-427.
- ↑ Triglycerides: Frequently Asked Questions, American Heart Association.
- ↑ M.G. Smith and M. Snyder, "Ethanol-induced virulence of Acinetobacter baumannii." 2005.
ReferencesISBN links support NWE through referral fees
- Boyce, John M., and Pittet Didier. “Hand Hygiene in Healthcare Settings.” Centers for Disease Control, Atlanta, Georgia, 2003. Retrieved March 19, 2022.
- Doxon, Lynn Ellen. The Alcohol Fuel Handbook. Tallgrass Research Center, 1980. ASIN B0006YPII4
- Lodgsdon, J.E. "Ethanol." In J.I. Kroschwitz (ed.). Encyclopedia of Chemical Technology, 4th ed. 9: 812–860. New York: John Wiley, 1994.
- Mathewson, S.W. The Manual for the Home and Farm Production of Alcohol Fuel. Berkeley, CA: Ten Speed Press, 1980. ISBN 0898150302
- McMurry, John. Organic Chemistry. 6th ed. Belmont, CA: Brooks/Cole, 2004. ISBN 0534420052.
- Morrison, Robert T., and Robert N. Boyd. Organic Chemistry. 6th ed. Englewood Cliffs, NJ: Prentice Hall, 1992. ISBN 0136436692.
- Sfetcu, Nicolae. Health & Drugs: Disease, Prescription & Medication. Lulu, 2014. ISBN 978-1312039995
- Smith, M.G., and M. Snyder. "Ethanol-induced virulence of Acinetobacter baumannii," 2005.
- Solomons, T.W. Graham, and Fryhle, Craig B. Organic Chemistry. 8th ed. Hoboken, NJ: John Wiley, 2004. ISBN 0471417998.
- Streitweiser, Andrew, Jr., and Clayton H. Heathcock. Introduction to Organic Chemistry. 4th ed. Pearson College Div, 1980 . ISBN 978-0139738500
External links
All links retrieved August 15, 2017.
- Sci-toys website explanation of US denatured alcohol designations.
- Reducing the negative effects of alcohol by taking cysteine and vitamin C.
- NIOSH Pocket Guide to Chemical Hazards.
- NIST Chemistry WebBook page for ethanol.
- Alcohol Specifications.
- ChEBI - biology related.
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.

![{\displaystyle \scriptstyle {\frac {[C_{2}H_{5}O^{-}][H^{+}]}{[C_{2}H_{5}OH]}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/4a1a83514d3f057bba95f26abcd385efd5b98a91)
![{\displaystyle \scriptstyle [H^{+}][OH^{-}]=1.0\times 10^{-14}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/a99b5f9b671eb27da64728d965b1c98ac4653a85)